Want Covid jab? Better be of age
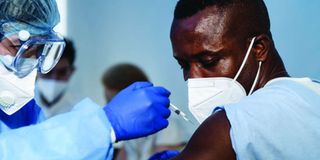
A medical staff injects Covid-19 vaccine on a man's arm.
What you need to know:
- Kenya says it will go on with procurement of Astra-Zeneca doses despite South Africa’s pause.
- New guidelines by World Health Organization recommend that those getting vaccines be aged at least 18
Interim guidelines by the World Health Organization for use of a vaccine against Covid-19 developed by Oxford University and AstraZeneca are discouraging vaccination of those aged below 18.
The guidelines, released yesterday, follow a trend in which most of the vaccine candidates, including those by Pfizer, Moderna and AstraZeneca, have been approved for emergency use in adult populations.
Pfizer’s vaccine has been authorised for use in ages 16 and above while Moderna’s offer is currently authorised for ages 18 and above. Both companies have begun clinical trials for younger populations.
This means there is currently no Covid-19 vaccine efficacy or safety data for children or adolescents below the age of 18 years.
“Until such data are available, vaccination of individuals below 18 years of age is not recommended,” warns the International Health Agency.
Prof Omu Anzala, a microbiologist based in Nairobi, said the nature of the pandemic is such that no one will be safe until everyone is safe. As such, many countries may not attain herd immunity when there are millions of children who are not immunised.
Children’s immune systems
In children, he said, the disease has very little adverse consequences compared to the rest. However, children are acting as a residual pool of hosts that are spreading a lethal disease to the general population.
“We need more trials on children since they have been harbouring non-Covid-19 coronaviruses for years,” said Prof Anzala.
Moderna has enrolled 3,000 adolescents to take part in a trial for those aged 12 to18 before embarking on an early childhood trial.
Trials involving children 11 years and younger will take longer, partly because the doses of vaccine will need to be de-escalated to lower strengths relative to the child’s weight, the firm said. This might affect its emergency roll-out since the approval may need to have a higher threshold.
Dr Walter Otieno, a paediatrician, said children’s immune systems are very different from adults’, and that their responses can be different at different ages, from infancy through teenage.
“Research on the Covid-19 vaccine for ages 16 and up needs to be repeated in children of younger ages. Until then, children should not be given the vaccines,” said Dr Otieno
“If we really want to mitigate harm or reduce transmission, then we need to start a trial on young children to test the vaccine effectiveness specifically in this population. They should not be left out.”
Covid vaccine jointly developed by Pfizer and BioNTech.
Pregnant women
In the interim, guidance developed on the basis of the advice issued by the Strategic Advisory Group of Experts on immunisation at its extraordinary meeting warned that pregnant women should receive the Astra-Zeneca vaccine only if the benefit of vaccination to the pregnant woman outweighs the potential risks.
For instance, if a health worker is at a high risk of exposure or has co-morbidities that place her in a high-risk group for severe Covid-19, then she should get the vaccine.
The WHO adds that the available data on the Astra-Zeneca vaccination of pregnant women are insufficient to assess efficacy or vaccine-associated risks in pregnancy.
Animal developmental and reproductive toxicity studies are ongoing, with preliminary findings showing no indication of harm to the development of the foetus.
Further studies are planned in pregnant women in the coming months, including a pregnancy sub-study and a pregnancy registry. As data from these studies become available, recommendations on vaccination will be updated accordingly, the advisory states.
Clinical trial
Pregnant women are at higher risk of severe Covid-19 compared with women of childbearing age who are not pregnant, and Covid-19 has been associated with an increased risk of pre-term birth.
The Oxford–AstraZeneca Covid-19 vaccine, codenamed AZD1222, is developed by Oxford University and AstraZeneca. It is an intramuscular injection, using as a vector the modified chimpanzee adenovirus ChAdOx1
Kenya is one of the countries, among them the UK, South Africa and Brazil, that are currently conducting clinical trials on the vaccine.
South Africa paused roll-out of the vaccine following a small clinical trial that showed it offered minimal protection against mild-to-moderate illness from the 501Y.V2 variant of the virus that is dominant in the country.
Kenya has reported that 24 million doses of the vaccine are expected in the country sometime next week.





